
The Regenerative Medicine Act came into effect on January 1, 2026, institutionally continuing regenerative medical technologies previously approved under the Regulations Governing Specific Medical Techniques (the “Special Approval Program”), thereby ensuring patient safety and protection of patient rights.
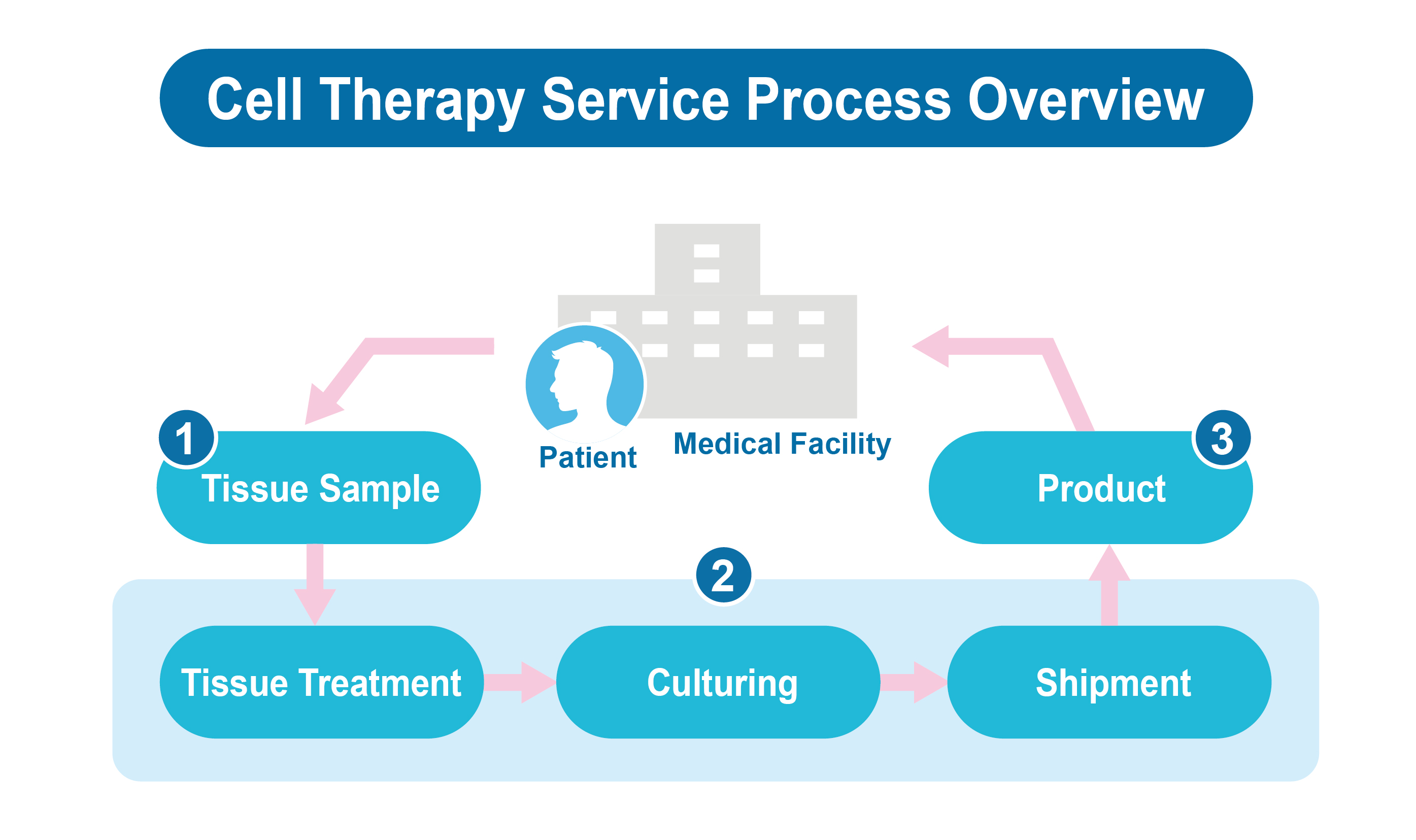
Ever supreme has long collaborated with multiple medical institutions to implement several regenerative medicine programs approved under the Special Approval Program, accumulating practical clinical experience and real-world data (RWD), which lay a solid foundation for regulatory transition and subsequent development under the framework of the Regenerative Medicine Act.
The Company focuses on the treatment of life-threatening or severely disabling diseases, in alignment with the policy objectives of the dual regenerative medicine laws. Leveraging its core technology platforms in immune cells, stem cells, and exosomes, Ever supreme continues to develop a diversified portfolio of regenerative medical technologies and regenerative medicine products, expanding treatment options within the current healthcare system and striving to improve patients’ quality of life.
| regenerative medical technologies | stage | approved medical institutions |
|---|---|---|
| ADCV01 | stage 1-4 | NTUH、CGH、TTZUCHI、CMUBH、TMANH |
| ADCV02 | stage 4 | CMUH、CMUHCH、TMANH |
| DC-CIK01 | stage 4 | CMUHCH、AUH、TMANH |
| CIK | stage 1-3 | SCMH、CBSCMH |
| CIK | hematoma | CMUH、KMUH |
| GDT | stage 4 | CMUH、TVGH、CMUHCH、AUH、KTGH、CCHCKB、TTZUCHI、TMANH、CMMC、TZUCHI |
| DC-CIK(WT1) | stage 4 | CMUHCH、TMANH、TZUCHI |
| BM-MSC | CMUH、CMUH-Taipei、CMUHCH、TMANH |
■ For information regarding autologous immune or stem cell therapy, please contact Ever-Supreme's official email address.: service@ever-supreme.com.tw
■ Or call us immediately for inquiries: 04-2325-2888
